Canon Medical begins development of rapid testing system for COVID-19
Mar 20, 2020
Share:
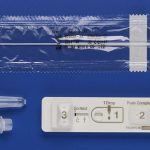
It seems that Fujifilm isn’t the only camera company making headlines in the fight against COVID-19 coronavirus. Canon has announced that Canon Medical Systems Corporation (Canon Medical) has started the development of a rapid genetic testing system for the novel coronavirus.
The rapid diagnostic test kits they say are part of a research programme focused on developing diagnostics methods for COVID-19 led by the Japanese Agency for Medical Research.
MELVILLE, NY, March 19, 2020 – Canon U.S.A., Inc., a leader in digital imaging solutions, today announced that Canon Medical Systems Corporation (Canon Medical) announced the start of development of a rapid genetic testing system for the novel coronavirus (COVID-19), reaffirming Canon Medical’s commitment to the basic research and development of rapid diagnostic test kits. This project is part of a research program focusing on the development of diagnostic methods for COVID-19 led by the Japan Agency for Medical Research¹.
Canon Medical was selected to participate in this research program in cooperation with Nagasaki University. This was in recognition of Canon Medical’s strength in leveraging its technologies in delivering practical solutions to support medical emergencies, notably by supplying Ebola rapid test kits to the Republic of Guinea in 2015², donating Ebola rapid test kits to the Democratic Republic of the Congo in 2019, and through the manufacturing approval and sale of the Genelyzer KIT (a reagent kit for Zika virus RNA testing) in 2018³.
The test and the reagents being developed for COVID-19 RNA testing is based on the LAMP method⁴ developed by Eiken Chemical Co., Ltd., and are to be used with a compact isothermal amplified gene fluorescent detector manufactured by Canon Medical to detect the presence of virus. Compared to the conventional test method of real-time PCR, the LAMP method allows for detection of the virus to be performed more easily and quickly, which makes it suitable for testing in local areas where infection is prevalent.
¹ On the AMED website: https://www.amed.go.jp/program/list/01/06/covid-19.html
² Following the outbreak of Ebola hemorrhagic fever in West Africa in 2015, in response to a request from the government of the Republic of Guinea, the Japanese government donated Ebola rapid test kits which were jointly developed by Canon Medical Systems Corporation (formerly Toshiba Medical Systems Corporation) and Nagasaki University.
https://www.mofa.go.jp/mofaj/press/release/press3_000088.html³ Trade name: Reagents for Zika Virus RNA Testing
Genelyzer KIT FGNK-0003A
Approval number: 23000EZX00035000
The company obtained approval for the manufacture and sale of this kit in June 2018 based on joint development work with Nagasaki University in two phases of the project “Establishing surveillance system for the risk of outbreaks of emerging and re-emerging insect-borne diseases in Japan and developing comprehensive insect-borne virus control system” (2016, 2017 onward) (research representative: Masahiro Hayashi, National Institute of Infectious Diseases of Japan) in the Research Program on Emerging and Re- emerging Infectious Diseases.
⁴ A nucleic acid amplification method known as LAMP (loop-mediated isothermal amplification) developed by Eiken Chemical Co., Ltd.
As to how soon Canon will be able to develop a fully working system or how quickly it might be deployed in Japan or elsewhere in the world upon completion is unknown, although judging by their response to the Ebola outbreak in West Africa in 2015, we may see some results fairly soon.
John Aldred
John Aldred is a photographer with over 25 years of experience in the portrait and commercial worlds. He is based in Scotland and has been an early adopter – and occasional beta tester – of almost every digital imaging technology in that time. As well as his creative visual work, John uses 3D printing, electronics and programming to create his own photography and filmmaking tools and consults for a number of brands across the industry.



































Join the Discussion
DIYP Comment Policy
Be nice, be on-topic, no personal information or flames.